Bridging the gap
MOLECULAR BRIDGES FOR HIGH-PERFORMANCE PRODUCTS

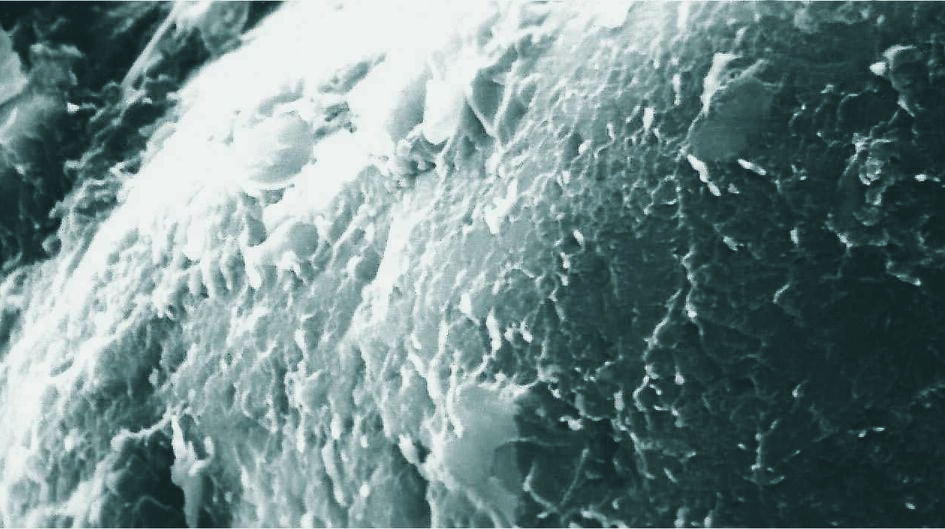


Inorganic minerals like kaolin, clay, talc, mica, or quartz are widely used as fillers in the thermoplastic or rubber composite industry. Silanes form permanent chemical bonds and therefore work as a molecular bridge between inorganic and organic elements which not only improve but create new functionalities.
Also, functional fillers like ATH (aluminium trihydrate) or MDH (magnesium hydroxide) as well as inorganic pigments like TiO2 are used to modify organic resins.
For this purpose, the mineral surface of the filler or pigment must be chemically modified in order to adapt it to the respective resin it will be incorporated in. Silanes are used for high quality surface modification. They chemically bond to the inorganic surface. Silane-induced filler-to-polymer bonding can actually be observed and monitored on a microscopic level. The treatment of fillers and pigments with Dynasylan® also provides processing advantages, for example:
- Improved filler dispersion
- Controlled rheological properties
- Improved wet-out between resin and filler
Dynasylan® MEMO acts as a molecular bridge to chemically bind the quartz into the resin. This molecular bridge cannot be mechanically destroyed resulting in a stable high-performance product. With no silane treatment, gaps exist between the filler surface and the surrounding polymer Matrix. These gaps are pre-determined fracture points in the filled polymer resulting in a low mechanical stability of the product.

